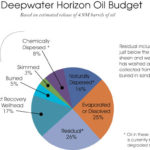
Hysteria gripped the nation while oil from BP’s well spewed into the Gulf of Mexico and oiled Louisiana’s marshes and beaches. Now, some Americans still refuse to eat seafood from this region. Are their fears valid?
In 1957, during the height of the Cold War, fiction writer Nevil Shute wrote On the Beach, a post-apocalyptic novel set in Australia shortly after World War III has wiped out all life in the northern hemisphere. With global air currents inexorably carrying the deadly fallout of nuclear war to the southern hemisphere, the novel’s characters set about trying to get in their last bits of enjoyment of “normal life” before radiation poisoning kills all of them.
In the first couple weeks after the blowout of the Macondo well, before extensive fishing areas were closed, an On the Beach mentality gripped Southeast Louisiana. Seafood restaurants and markets experienced an almost mad run of consumers set on getting a last meal of seafood. Recreational anglers rushed to get in as many fishing trips as possible, not knowing when they might be able to go again.
Then areas began to be closed in ever-expanding concentric rings around the spill site. Supplies of Louisiana seafood became tight to non-existent, and recreational fishermen parked their boats, covered them in tarps and looked away.
Fear of the unknown set in. A cascade of doomsday predictions by environmental groups transmogrified the fear of a public distrustful of their government and already scared by decades of negative publicity about their food, water and air into paranoia.
A peek at the language used to describe the spill illustrates why people were frozen in fear: “environmental catastrophe,” “apocalypse,” “toxic shock,” “toxic illnesses,” “highly toxic to marine life,” “badly contaminated,” “biodiversity disaster,” “notoriously toxic,” “environmental tragedy,” “toxic chemicals,” “environmental minefield,” “toxic cocktail” and “irreparable damage.”
The use of dispersants was described as a “giant chemistry experiment,” an “eco-toxicological experiment” and a “draconian experiment.”
Lost in the escalating rhetoric was the perspective of experience. In 1979, Ixtoc I, near Campeche, Mexico, blew out and ran oil into the Gulf of Mexico for 9 months, ultimately discharging 3.5 million barrels of oil and fouling 162 miles of Texas beaches with oil up to a foot deep in some places. During the blowout, 493 aerial missions were flown to dump Corexit 9527 dispersant on the crude oil.
Scientists found little damage in the aftermath of the Ixtoc I blowout. Luis Soto, a marine biologist from the National Autonomous University of Mexico said, “This is not to say there were no consequences — just that evidence is that these are not as dramatic as we feared. After about two years, the recuperation was well on the way.”
As a matter of fact, Campeche shrimp harvests returned to pre-spill levels during that period of time.
A report prepared by the U.S. Bureau of Land Management on the effects of the spill on U.S. waters concluded, “In spite of a massive intrusion of petroleum hydrocarbon pollutants from the Ixtoc I event into the study region of the South Texas Outer Continental Shelf during 1979-1980, no definitive damage can be associated with this or other known spillage events (e.g., Burmah Agate) on either the epibenthic [living on the bottom] commercial shrimp population (based on chemical evidence) or the benthic infaunal [all bottom life] community.”
Burmah Agate, as referred to in the report, was a ship collision southeast of Galveston, Texas, in 1979 that released 2.6 million gallons of oil into the Gulf.
Most succinct was Olof Linden of the World Maritime University in Malmö, Sweden, and a member of a United Nations group of experts that assessed the spill: “We were surprised there were so relatively few effects.”
Some public statements made by scientists, environmentalists and journalists about the BP spill were incorrect. Many others, especially those made by scientists, were couched in terms of what could happen or may happen.
“That’s what scientists do,” says Dr. Ed Overton, environmental chemist and professor emeritus at the Louisiana State University Department of Environmental Science. “Scientists try to consider everything possible. What the public hears is ‘Oh my gosh, these things could happen — these things are going to happen.’ What is possible is not always probable.
“The Gulf of Mexico could get hit by an asteroid, again. [Scientists generally agree that what is now the Gulf of Mexico was hit by an asteroid 4-9 miles in diameter at the end of Cretaceous Period 65 million years ago, causing mass extinction of dinosaurs.] It’s possible, but is it probable? No, it’s not probable. Scientists have to be careful about using ‘possible’ and ‘probable.’
“Part of it [misstatements and misinformation] was people talking about oil spills who didn’t know anything, no background or experience, about oil.
“Also, very clearly, people were trying to use this event to push a political agenda to stop offshore drilling and push change to a hydrocarbon-free energy supply. There was a certain amount of effort to take advantage of the disaster to make it sound like it was worse than it was.”
Fear-engendering statements using could, may and might were rife in print, broadcast and electronic media. So were outright incorrect statements.
“The crude gushing from the well contains vast amounts of natural gas that could pose a serious threat to the Gulf of Mexico’s fragile ecosystem” (journalist).
“Huge quantities of methane have entered the Gulf, scientists [unidentified] say, potentially suffocating marine life….” (journalist).
“It could take years, possibly decades, for the system to recover…” (scientist)
“Crude oil could enter a current that would draw it out of the Gulf and up along the East Coast…” (scientist).
“Marine life may take decades to recover, wiping out businesses along the coastline that depend on the fishing and seafood industry” (journalist paraphrasing a scientist).
“Scientists [unidentified] say oil could stunt the animals’ [sea turtles] growth….” (journalist).
“Getting toxicity into that part of the food chain is a real problem…” (inaccurate statement by environmentalist).
“There are fears that an entire year’s worth of bluefin tuna larvae may have perished” (blogger).
“Corexit is in the water column, just as we thought, and it is entering the bodies of animals. And it’s probably having a lethal impact there” (inaccurate statement by environmentalist).
“The BP drilling disaster is having an untold impact on the amazing marine life of the Gulf of Mexico” (inaccurate statement by environmentalist).
“To make things a little scarier, some of the oil-eating bacteria have been genetically modified or otherwise bio-engineered to better eat the oil…” (inaccurate statement by environmentalist/scientist).
“The devastation could last ‘years or decades. It looks like everything is dead’” (journalist quoting a scientist).
“Corexit dispersants, in combination with crude oil, pose grave health risks to marine life and human health and threaten to deplete critical niches in the Gulf food web that may never recover” (environmentalist).
“We have dolphins that are hemorrhaging. People who work near it are hemorrhaging.” (39-year career EPA whistleblower).
“And then we are likely further damaging a fragile ecosystem [by using dispersants] in the name of saving it, and imperiling the health of seafood eaters for years to come” (blogger).
Paranoia in the face of such an “informational” blizzard isn’t surprising. On May 18, 2010, the following panicky comment was left on a blog: “Just thinking….. Has anyone brought up the possibility of the crude oil-dispersant mix being pulled up into the vortex of hurricanes, and being thrown far and wide into the upper atmosphere, and for those same chemicals to rain down upon us, upon the earth’s animals and plants?”
Late in the spill and after the capping of the MC 252, much of the attention of environmental activists was shifted to human health. Dr. Riki Ott, who always identifies herself as an “Exxon Valdez survivor” and “environmental activist” and alternatively a “marine biologist” or “marine toxicologist,” has led charges that oil-eating bacteria have turned to assaulting humans and that dispersant chemicals are “busting red blood cells.”
She proclaimed that, “The people, particularly in Grand Isle, have been poisoned by tyrants that care more about their pocketbooks than human life or mother earth,” adding “people (are) now dropping dead,” in the Gulf region.
The Louisiana Department of Health and Hospitals Section of Environmental Epidemiology & Toxicology tracked data from hospital emergency departments, outpatient clinics, doctor’s offices and the Louisiana Poison Center from the week of April 18 through the week of Sept. 19, two months after the well was capped. The monitoring found 415 reports of health complaints believed to be related to exposure to the oil spill. Clean-up workers reported 329 of these, and 86 came from the general public of which 78 reported being exposed in their homes.
The summary concluded, “Most frequently reported symptoms include headache, dizziness, nausea, vomiting, weakness/fatigue and upper respiratory irritation. One hundred sixty-nine workers had heat-related complaints. Eighteen had short hospitalizations. The general public complaints were primarily related to odors with mostly mild symptoms being reported.”
When tests by state and local agencies consistently showed the amounts of oil-spill pollutants in fish and seafood to be well below “levels of concern,” interest groups attacked figures that were used for seafood consumption in Louisiana. In a December release, New Iberia environmental activist and biochemist Wilma Subra said, “I’ve been requesting that we redo the calculations and adapt to the dietary habits of people on the coast.”
Peter Brabeck, an environmental monitor for the Louisiana Bucket Brigade, added, “The (Food and Drug Administration) protocol is absolutely 100 percent inadequate. No question about it.”
In the release, the Natural Resources Defense Council released a non-scientific survey of their own questioning the amounts used by the FDA, Environmental Protection Agency and NOAA to determine level of concern. NRDC senior scientist Gina Solomon accused FDA of setting safety standards for cancer-causing chemicals based on nationwide consumption rates rather than “the uniqueness of the regional diet.”
Most scientists charged with dealing with fish and seafood safety in the aftermath disagree with that assessment. Marine biologist Dr. Bob Dickey, FDA director of the Gulf Coast Seafood Laboratory, said the figures used are “very protective,” and “grounded scientifically in the national survey process by the Centers for Disease Control and the National Institutes of Health, a very well-established and validated process.
“We have to have a validated scientific foundation. We first discarded all non-seafood-eaters from the group. Then we decided to take the 90th percentile of seafood eaters [the top 10 percent]. I am comfortable with the consumption rates.
“I am concerned about the distrust engendered by some parties. The picture that is being presented is that we are holding something back, and we are not. It is disconcerting when you see the accusations.”
In response to the charges over seafood-consumption figures not providing a margin of safety, LDHH re-evaluated their results based upon the chemical analyses for edible seafood tissue through early February 2011. The results show that an individual could eat 9 pounds of fish, 5 pounds of oysters or 63 pounds of shrimp and crabs per day without exceeding established acceptable health risk levels from oil-spill pollutants.
Dr. Jimmy Guidry, the Louisiana State Health Officer and Medical Director for LDHH says flatly, “As a physician, I totally support the fact that eating seafood is not going to be unsafe. I eat the seafood. My grandchildren eat the seafood. There is no doubt in my mind that the seafood is safe to eat.”
Olivia Watkins, Executive Media Advisor for the Louisiana Department of Wildlife and Fisheries, offers perspective.
“I suspect that some groups feel it is their responsibility to challenge science,” she said. “However, it is critical to examine science. When you hear a message, evaluate who is presenting, whether it’s a private organization or a public agency, and why.”
Dr. John Bell, professor in the LSU AgCenter’s Department of Food Science, is blunter.
“Every individual is concerned when an interest group brings up concerns,” he said. “The public should question whether they [the interest groups] have agendas and if they are neutral and independent. Or do they have an interest in scaring the public?
“Attorneys are obvious. NGOs [non-government organizations] that are seemingly there to protect the public may be trying to grow something out of proportion for the financial benefit of their organizations.
“A recent example is mercury. Fears of natural low levels of background mercury in oceanic fish were fanned to create fear. Actually, oceanic fish are high in selenium, which binds mercury and makes it biologically inactive. It can’t pass the blood-brain barrier. Mercury could be found in human hair and blood, but it was bound mercury.
“The loss of benefits from not eating fish was a worse health impact than eating them. It is an example of groups using partial science to fan fears and food health concerns to further their own agenda.”
A chemistry lesson
“It is often said that we don’t know what is in oil. Ditto for dispersants,” says LSU’s Overton. “The truth is that we do know and we have been knowing. What was said was said to grab people’s attention.
“Oil is a strange thing. It is physically a liquid with a slimy texture, but it doesn’t boil and it doesn’t have the ability to mix with water. It has thousands of different molecular sizes and shapes.
“It started off as biomass, the materials of life, which were buried by sediment. It cooked under pressure and converted to liquid hydrocarbons. If it cooked too much, it became gas, mostly methane, but also ethane, propane and butane.”
Dr. Ralph Portier, a professor of Environmental Sciences whose specialty within the LSU School of the Coast and Environment is marine microbiology and toxicology, also calls crude oil a mixture of “hundreds of chemical species” that are “biogenic,” meaning that they were formed under pressure from primitive plant life from either land or the sea.
Portier explains that three criteria are used to describe this mixture of hydrocarbons: whether it is sweet or sour, light or heavy and how much energy value it has. Sweet crude has little sulfur content. Light crude is made up of a higher percentage of lighter compounds and is easier to pump, refine or biodegrade than heavy crude. South Louisiana crude oil is light, sweet crude.
“BP oil is classic South Louisiana crude oil,” he said, “with the caveat that it has just a little bit more asphalt than the ‘honey sweet’ crude that we have been pumping since the 1930s. Analytically, we can fingerprint oil close enough to tell Gulf of Mexico Oil from inland [Louisiana] oil. BP oil is older than inland oil, but analytically has a very, very similar signature.”
Dr LuAnn White, a toxicologist with the Tulane University School of Public Health and Tropical Medicine, director of the Center for Applied Environmental Health and a diplomat of the American Board of Toxicology, breaks down the chemistry of oil in more detail. Oil molecules, she explains, can be grouped into four categories by the number of carbon atoms they contain and, therefore, the size of their molecules.
C1 to C4 molecules are gases such as natural gas, propane and butane, and have no direct chemically toxic health effects other than asphyxiation by displacement of oxygen in confined spaces. They may compose up to 40 percent of the leaking crude.
C5 to C16 molecules are volatile liquids that have very high evaporation rates. They come in two forms. Aliphatic compounds have their carbon atoms arranged in chains. The carbon atoms of aromatic compounds are arranged in rings. Aromatic compounds contain the most toxic fraction of crude oil, including benzene, toluene, ethyl benzene and xylenes, abbreviated collectively as (BTEX).
Benzene has carcinogenic properties, and the others cause irritation of the lungs, eyes, and skin and mucus membranes. They do not bioaccumulate in animal flesh, and if eaten, are rapidly metabolized and passed out of the body. All are in gasoline, and people are exposed to them when they fuel their automobiles. Analysis of surface oil from the BP spill has revealed no BTEX.
C16 to C22 molecules are called heavier liquids. They may be aliphatic, with their carbon atoms arranged in chains or found with multiple interlocking rings as polycyclic aromatic hydrocarbons (PAHs). Aliphatics have very low toxicity to humans and cause damage mainly by physically coating and incapacitating aquatic life and birds.
At least 39 PAHs are reported from petroleum analyses. They are not absorbed well into the body, do not bioaccumulate and are excreted rapidly, especially by fish and other vertebrates and, to a lesser degree, by crustaceans such as shrimp and crabs. Excretion is lower in mollusks, such as oysters. Some PAHs are carcinogenic, and since they stick around longer than many other oil molecules before degrading, they are what seafood is tested and monitored for.
Oil in this third group forms what was most commonly seen in Louisiana waters as “mousse,” a thick, gunky emulsion of oil and water. Some but not all samples of mousse have PAHs in them. When the source of oil disappears, PAHs return to background levels in weeks.
C22 to C25+ molecules are the heaviest components of crude oil. These are the asphaltenes, parafins and tars. They are practically non-toxic, but are slow to break down and, therefore, messy and ugly. Tar balls are a typical example.
Portier points out that about 98 percent of the oil in the BP spill was aliphatics and a half of a percent was asphalt, leaving only 1 to 2 percent as aromatics. Of these, PAHs were only a small subset, in the parts per million.
The chemistry of dispersants is relatively simple. Overton calls dispersant a “fancy name for soap,” with ingredients used in foods, pharmaceuticals and around the house. White also likens them to household soaps used to break up oil from a frying plan, allowing it to be washed out.
Corexit 9500 contains three components — a surfactant, petroleum distillates and propylene glycol. Depending upon conditions, it has a half-life of two days to two weeks. In other words, some time between two days and two weeks, half of the applied dispersant has broken down. Two days to two weeks later, half of what remains has broken down, and so forth. No dispersants were applied in the Gulf of Mexico after July 19.
The surfactant in Corexit is sodium dioctylsulfosuccinate, known as DOSS. Since this ingredient is the slowest to break down, it has been identified as the best one to track the presence of the dispersant. To keep things in perspective, FDA notes that DOSS is an acceptable ingredient in beverages at levels up to 10 parts per million, and that it is used in pharmaceuticals and in stool softeners available over-the-counter in dosages as high as 250 mg per capsule. NOAA and FDA scientists, as well as White, Overton and Portier, agree that it does not bioaccumulate in the food chain.
Dispersants do not change the amount of oil in the water directly, but break it into small droplets that are more easily degraded by natural microorganisms in the water.
EDITOR’S NOTE: In the May issue, we’ll look at the “toxicity” of oil as well as how Gulf seafood is tested in the wake of the spill.